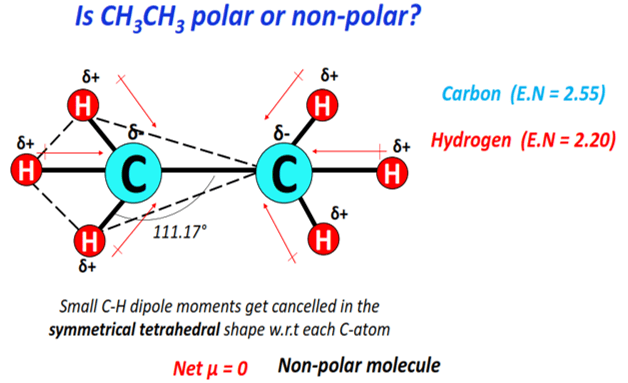
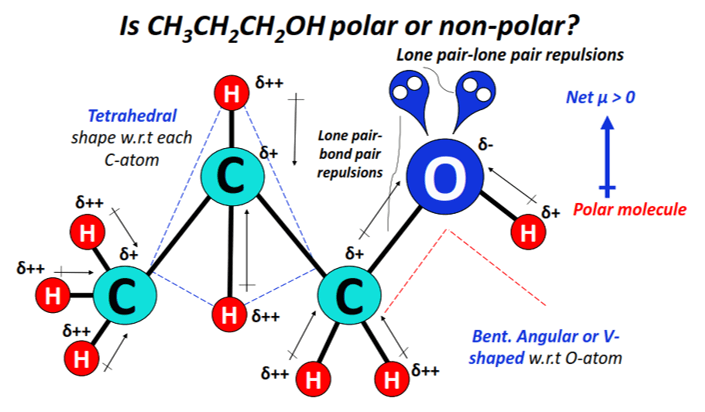
C2H6O Lewis Structure (Dimethyl Ether) | C2H6O Lewis Structure (Dimethyl Ether) Several people get confused when they look at the chemical formula of C2H6O as it is a general formula for both...

SOLVED: 1. There are three structural/constitutional isomers of C2H6O. Draw all three isomers. 2. Considering bond polarity and molecular shape, write whether each isomer is polar or nonpolar. a. If you need

Is CH3NH2 polar or non-polar? - Polarity of CH3NH2 | Molecular geometry, Covalent bonding, Electron configuration

Is n pentane an ionic molecular nonpolar or molecular polar compound what intermolecular forces are present? - Opera Residences

Alcohols are organic molecules that have a hydroxyl (-OH) group that is connected by a single bond to a carbon atom. Let's consider the molecule ethyl alcohol (C2H6O). a. What is the




![Biology] Polar Bonds - General Discussion - Neowin Biology] Polar Bonds - General Discussion - Neowin](https://cdn.neowin.com/forum/uploads/post-107847-1188869267_thumb.jpg)